When Other Treatments Have Failed, There Is Hope
Harmony is proud to partner with NeuroStar to provide transcranial magnetic stimulation (TMS) as a treatment option for our clients. It is FDA-cleared to treat depression, depression with anxiety, and OCD. Most major insurance plans cover NeuroStar® TMS, including Medicare and Tricare plans nationwide.
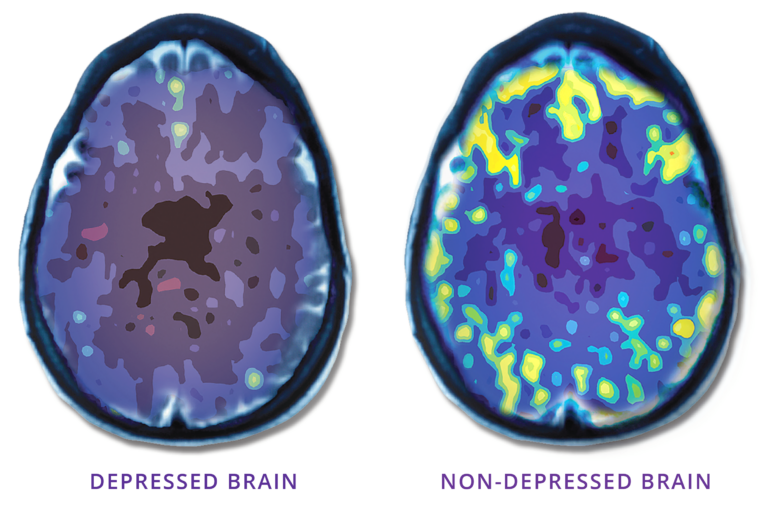
TMS uses focused magnetic pulses, similar in strength to an MRI, to revitalize underactive areas of the brain involved in regulating mood. It is not ECT (electroconvulsive therapy), “shock therapy,” or surgery.
How NeuroStar TMS Can Help
NeuroStar® TMS has helped thousands of people with depression get relief from their depression symptoms.
6.9
million NeuroStar Advanced TMS treatments have been performed in over 188,000 patients
83%
of patients experience an improvement in the severity of their depression
62%
of patients show complete remission
Neurostar TMS can offer hope to those who have struggled to find relief from their depression symptoms.
If you have been diagnosed with major depressive disorder (MDD) and have not experienced relief from antidepressants, NeuroStar TMS might be right for you. NeuroStar TMS treatment is also covered by insurance for more than 300 million people.
How NeuroStar TMS Therapy Works
TMS treatment is conducted in one of our Harmony clinics. You will meet with our team for a consultation to determine your eligibility. Depending on your provider’s recommendation, a typical TMS treatment plan includes 36 sessions that each last between 19–37 minutes. Completing the full treatment course gives the best chance for delivering long-lasting relief from depression.
- Before treatment – You’ll recline comfortably in the treatment chair. A curved magnetic coil will be positioned on your head.
- During treatment – You’ll hear a clicking sound and feel a tapping sensation on your head as the NeuroStar coil focuses treatment precisely at the source of the depression: the brain. The patented Contact Sensing technology ensures the prescribed dose is delivered, every treatment, every pulse.
- After treatment – With NeuroStar TMS, you are fully awake and alert during treatment and may drive home and resume daily activities immediately afterward. There are no negative effects on memory or sleep.
With Harmony, you’re supported every step of the way on your journey to regain control and live a fulfilling life free from the constraints of depression.
Find out if NeuroStar TMS is right for you.
Frequently Asked Questions
NeuroStar TMS uses focused magnetic pulses (similar in strength to an MRI) to revitalize underactive areas of the brain involved in regulating mood. When these regions are underactive, depression can result. “Waking up” these regions can have a lasting effect on depression, making long-term remission from depression a reality for many people.
A real-world study reported an 83% response rate. This means that 83% of patients who completed their NeuroStar TMS treatment cycle saw measurable improvement in their depression symptoms. The same real-world study reported a 62% remission rate. This means that 62% of patients who completed their NeuroStar® TMS treatment cycle likely wouldn’t be diagnosed with MDD if they were first being evaluated by their doctor.
Yes, TMS is a safe and FDA-cleared treatment for depression, depression with anxiety, and OCD. Over 6.9 million NeuroStar Advanced TMS treatments have been performed in over 188,000 patients, and less than five percent of clients reported any side effects. The most common side effect is a headache right after TMS treatment, which goes away after the first week of sessions for most people.
Many patients experience side effects from their medication, increasing the chance that they will stop taking them. The most common side effect with NeuroStar is mild to moderate pain or discomfort at the treatment site. For most patients, this subsides within the first week of treatment.
Most major insurance plans cover NeuroStar TMS, including Medicare plans nationwide. Co-pay assistance and payment plans are also available. Our team at Harmony can help you check coverage and discuss co-pay costs.
Adults aged 22-70. Adolescents aged 15-21 are eligible for TMS as an add-on therapy.
NeuroStar TMS is FDA-cleared for the treatment of major depressive disorder (MDD) in adults 22-70, as an add-on therapy for adolescents aged 15-21, and for the treatment of obsessive-compulsive disorder in adults aged 22-70.
Adolescents and TMS Therapy: A Path to Healing and Hope
NeuroStar TMS is a promising option, available to adolescents aged 15-21 as an add-on therapy for treatment-resistant depression (TRD). Depression can significantly impact adolescents, affecting their daily lives and future. This is why we are thrilled to extend the benefits of NeuroStar Advanced TMS therapy to adolescents who struggle with TRD.
NeuroStar Advanced TMS therapy offers an effective and innovative non-invasive treatment option, distinct from traditional depression therapies and free from medication-related side effects. Supported by clinical research, it demonstrates significant improvement in depressive symptoms, promoting long-term mental wellness for adolescents seeking relief.
For young patients who have not had success with other depression treatments, TMS offers a new path to healing. We encourage parents and caregivers to fill out the form on this page to learn how NeuroStar Advanced TMS can improve their loved one’s life.
Find out if NeuroStar TMS is right for you.
Call Harmony for Depression Relief Today
At Harmony, we are proud to offer a full suite of mental health services, including therapy and psychiatric medication management and alternative treatment options for depression like NeuroStar TMS and Spravato. This collaborative, multidisciplinary approach allows our team to create an individualized treatment plan that is tailored to you.
Call us today at 304.410.0082 to find out if TMS therapy is suitable for you or your loved one. You can also complete our TMS form below to determine if you or your loved one is a good candidate for NeuroStar TMS.
Indication Statement
The NeuroStar Advanced Therapy System is indicated for the treatment of depressive episodes and for decreasing anxiety symptoms for those who may exhibit comorbid anxiety symptoms in adult patients suffering from major depressive disorder and who failed to achieve satisfactory improvement from previous antidepressant medication treatment in the current episode.
The NeuroStar Advanced Therapy system is intended to be used as an adjunct for the treatment of adult patients suffering from obsessive-compulsive disorder.
NeuroStar Advanced Therapy is only available by prescription. A doctor can help decide if NeuroStar Advanced Therapy is right for you. Patients’ results may vary.
The most common side effect is pain or discomfort at or near the treatment site. These events are transient; they occur during the TMS treatment course and do not occur for most patients after the first week of treatment. There is a rare risk of seizure associated with the use of TMS therapy (<0.1% per patient).
Visit NeuroStar.com for full safety and prescribing information.
Clinical and Academic References
- Carpenter LL, et al. (2012). Transcranial Magnetic Stimulation (TMS) for Major Depression: A Multisite, Naturalistic, Observational Study of Acute Treatment Outcomes in Clinical Practice. Depression and Anxiety, 29(7):587-596. www.ncbi.nlm.nih.gov/pubmed/22689344
- George MS, et al. (2010). Daily Left Prefrontal Transcranial Magnetic Stimulation Therapy for Major Depressive Disorder: A Sham-Controlled Randomized Trial. Arch Gen Psychiatry, 67(5):507-516. www.ncbi.nlm.nih.gov/pubmed/20439832
- Dunner DL, et al. (2014). A Multisite, Naturalistic, Observational Study of Transcranial Magnetic Stimulation (TMS) for Patients with Pharmacoresistant Major Depressive Disorder: Durability of Benefit Over a 1-Year Follow-Up Period. J Clin Psychiatry, 75(12):1394-1401. www.ncbi.nlm.nih.gov/pubmed/25271871
- O’Reardon JP, et al. (2007). Efficacy and Safety of Transcranial Magnetic Stimulation in the Acute Treatment of Major Depression: A Multisite Randomized Controlled Trial. Biol Psychiatry, 62(11):1208-1216. www.ncbi.nlm.nih.gov/pubmed/17573044

